What Are Surfactants?
Surfactants are a category of chemical compounds that are used in lowering the surface tension (or interfacial tension) between different compounds, such as two liquids or between a gas and a liquid, or it can also be between a liquid and a solid. Surfactants are categorised as organic compounds and are amphiphilic in nature. It basically means that they contain both hydrophobic and hydrophilic groups.
In other words, a surfactant has both a water-insoluble component and a water-soluble component. One of the common properties of surfactants is that they will diffuse in water and adsorb at interfaces between air and water. It can also adsorb at the interface between oil and water where water is mixed with oil. The water-insoluble group can extend out of the bulk water phase and move into the air or oil phase. On the other hand, the water-soluble head group usually stays in the water phase.
The word surfactant has been derived from the word surface active agent and was coined in the year 1950. In any case, surfactants today are found in many products that we use today. They are found in detergents, wetting agents, foaming agents, emulsifiers and dispersants. Surfactants are one of the important components in detergents that help to eliminate dirt from clothes, skin and household utensils, mainly in bathrooms and kitchens. Besides, they are extensively used in industries.
Soaps are the foremost surfactants, mostly obtained from fats called glycerides, which are mainly esters formed by trihydric alcohol, glycerol, with fatty acids having lengthy chain carboxylic acids. Glycerides are hydrolysed by heating with a solution of sodium hydroxide to make soaps, sodium salts of acids and propane 1,2,3-triol. This process is called saponification.
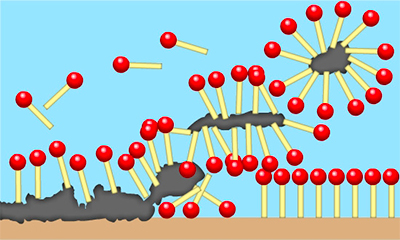
How Do Surfactants Work?
When a sufficient amount of surfactant molecules are added to a solution, they start combining together. During this, they form structures or aggregates called micelles in the bulk aqueous phase. As the micelle starts to form, the surfactant heads (hydrophilic heads) remain exposed to water or the surrounding liquid. The tails (hydrophilic heads) come together at the centre of the structure and remain protected from water. Different types of aggregates, such as spherical or cylindrical micelles or lipid bilayers, can also be formed. Furthermore, the shape of the aggregates mostly depends on the chemical structure of the surfactants (balanced size between the hydrophilic head and hydrophobic tail).
In any case, surfactants work by breaking down the interface between oils, water or dirt. The oils and dirt are also held in suspension, making it easy to remove them.